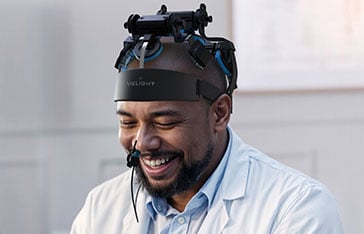
- Vielight Neuro 4 User Guide
Video Tutorial | Informational Video - Vielight Neuro Pro 2 Black
Setup Guide | Video Tutorial | Informational Videos | Practitioner’s Guide | App Guide - Vielight Vagus
User Guide | Video Tutorial | Informational Video - Vielight X-Plus 4
User Guide | Informational Video - Vielight MIP
User Guide - Vielight Neuro Pro 2 White
User Guide
- Do Vielight devices penetrate the skull and skin?
Yes. Watch a demonstration from the Vielight Lab. - Do Vielight Neuro devices provide full transcranial coverage?
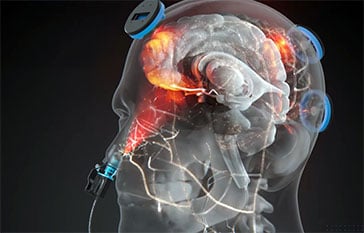
Yes and more. Bone and skin scatters light energy naturally. With just 5 powerful Vie-LED modules, we have full transcranial coverage with a weighted geometry on the hubs of the Default Mode Network.
Additionally, our patented intranasal technology allows NIR energy to reach deep brain structures, otherwise inaccessible from the transcranial side. The Vielight Neuro is the only brain photobiomodulation device in the world that provides comprehensive coverage on all sides of the brain through our patented intranasal-transcranial photobiomodulation (itPBM) process.
Watch the Vielight Neuro’s patented full intranasal-transcranial coverage on a real human skull with a CMOS camera: https://www.youtube.com/watch?v=Onm07-EPCkw - Is the Vielight Neuro a near-infrared helmet?
Yes, the Vielight Neuro is a next-generation NIR helmet specifically engineered for brain photobiomodulation (PBM). While traditional helmets often suffer from hair blockage and low power, the Neuro utilizes industry-leading irradiance, concave LED modules designed to optimize scalp contact.
The Neuro provides full transcranial coverage by leveraging light scattering. This ensures that the NIR energy is distributed effectively across the cortex, with a specific focus on the Default Mode Network (DMN)—the brain’s primary functional network.
Unlike basic helmets, the Neuro also includes a patented intranasal applicator. This allows the system to reach deep ventral brain structures that are otherwise inaccessible by transcranial-only helmets. https://www.vielight.com/blog/understanding-the-vielight-neuro/ - What is intranasal-transcranial photobiomodulation?
Intranasal-transcranial photobiomodulation is Vielight’s patented Intranasal-transcranial photobiomodulation is Vielight’s patented technology that combines light delivery through both the nasal cavity and the scalp to stimulate the brain.
Specifically, it uses near-infrared (NIR) light for full-transcranial coverage with a focus on the brain’s Default Mode Network (DMN) and deep ventral structures that are otherwise difficult to reach through the skull alone.
–Transcranial: High-irradiance modules provide full transcranial stimulation of the cortex, focusing on the nodes of the Default Mode Network (DMN).
–Intranasal: A specialized applicator reaches the ventral brain structures (the underside of the brain) through the cribriform plate.
By combining these two pathways, itPBM provides a more comprehensive brain stimulation profile than standard transcranial photobiomodulation helmets (tPBM).
Read Baycrest’s study highlighting itPBM 20x efficiency advantage: https://www.vielight.com/blog/baycrest-hospital-vielight-pbm-improves-brain-fluid-dynamics-mri/
Medical Device User Guides
- Vielight RX-Plus User Guide | Link
How long does each session last?
Each Vielight device is auto-timed for ease of use:
- Intranasal devices: 25 minutes per session
- Vielight Neuro (Alpha, Gamma, Duo): 20 minutes per session
- X-Plus 3: 25 minutes per session
- MIP: 25 minutes per session (recommended twice daily)
How often should I use my Vielight devices?
Neuro Series (Alpha, Gamma, Duo)
The Vielight Neuro is designed for safe, effective home use with simplicity in mind. We recommend the following general guidelines:
- Alpha: Ideal for general brain wellness, stress recovery, relaxation, and improving sleep quality.
- Gamma: Best used for tasks requiring focus, creativity, cognitive support, or deeper meditation. Note: Some users may find Gamma too stimulating before bedtime.
Suggested Usage Pattern for Neuro Duo:
- Use Alpha on Day 1, Gamma on Day 2, and continue alternating for six days. Take a full rest day on Day 7 to allow for integration
Vielight X-Plus 3
- Recommended once daily (auto-timed for 25 minutes)
Vielight MIP
- Recommended twice daily: once in the morning and once in the evening
General Guidelines (All Devices)
- Healthy brain: Use your device once every two days
- Needing support: Use once per day, up to 6 days per week
If you’re unsure about your routine, feel free to contact us for guidance.
Can I use two devices in each nostril at the same time?
Yes, you can. For example, you may use the Vielight 633 or 655 in one nostril and the Vielight 810 in the other simultaneously. These wavelengths target different systems and do not interfere with each other.
A research department at the U.S. Veterans Affairs uses this method in clinical settings. (Link to reference)
Does it matter which nostril I use?
Not at all. Either nostril can be used.
However, if you have sinus or eye-related issues, we suggest alternating nostrils occasionally to stimulate both sinuses, optic nerves, and eyes.
Can children use Vielight devices?
Yes, children can use the devices, but we recommend starting with shorter sessions (5–10 minutes) and gradually increasing time as tolerated.
If headaches or discomfort occur, reduce the session time.
The Vielight Neuro was used in an Autism clinical trial on children with age ranges between 5-15 years old.
Can I share a device with someone else?
Yes, sharing is possible. However, it is strongly recommended to disinfect the intranasal applicator between uses with an alcohol swab.
Alternatively, separate nose applicators can be purchased for personal hygiene.
Alzheimer’s Disease (Dementia) / Mild Cognitive Impairment (MCI)
- Effects of Home Photobiomodulation Treatments on Cognitive and Behavioral Function and Resting-State Functional Connectivity in Patients with Dementia: A Pilot Trial
Institutes – University of California San Francisco & Veterans Affairs USA
Published Study | Summary | Device: Vielight Neuro Gamma - Vielight Neuro RX Gamma – Alzheimer’s Disease Pivotal Phase III Clinical Trial (n=228)
Co-institutes – St Michael’s Hospital, Toronto
Clinical Trial Information Link (228 participants | 3 years | Ongoing) - Revitalizing Cognition in Older Adults at Risk for Alzheimer’s Disease With Near-Infrared Photobiomodulation
Institutes – University of Florida, University of Arizona, National Institute on Aging (NIA)
Clinical Trial Information Link (168 participants | 5 years | Registration) - Significant Improvement in Cognition in Mild to Moderately Severe Dementia Cases Treated with Transcranial Plus Intranasal Photobiomodulation: Case Series Report
Co-authoring institutes – Harvard Medical School, Boston University School of Medicine
Published Study - Impact of Photobiomodulation (PBM) on Biomarkers of Alzheimer’s Disease
Institutes – University of California San Francisco
Clinical Trial Information Link / UCSF Clinical Trials (16 participants | 1.5 years | Ongoing) - A Pilot Study Evaluating the Feasibility, Safety, and Efficacy of Transcranial Photobiomodulation (tPBM) for the Treatment of Mild Cognitive Impairment (MCI)
Published Presentation
Traumatic Brain Injury / Concussion
- Transcranial Photobiomodulation Treatment Effects In Former Athletes With Repetitive Head Hits (n=49)
Institutes – Department of Neurology, University of Utah
Published Study | Summary | Device: Vielight Neuro Gamma (49 participants) - A Proof-of-Concept Study Investigating the Effects of Transcranial Plus Intranasal Photobiomodulation on Cognitive Function after Repetitive Head Acceleration Events
Institutes – Department of Neurology, University of Utah
Summary | Published Study (45 participants) - Exploring a New Frontier in First Responder Wellness: Transcranial-Intranasal Photobiomodulation
Summary | Published Study (15 participants) - Transcranial Photobiomodulation Treatment: Significant Improvements in Four Ex-Football Players with Possible Chronic Traumatic Encephalopathy
Institutes – Boston University School of Medicine, Harvard Medical School
Published Study Link - Changes in Brain Function and Structure After Self-Administered Home Photobiomodulation Treatment in a Concussion Case
Institutes – VA Advanced Imaging Research Center, San Francisco VA Health Care System, University of California, San Francisco
Published Study Link (Frontiers, Neurology, 2020) - Photobiomodulation to Improve Cognition in TBI, With fMRI
Institutes – Boston Veterans Affairs Research Institute
Clinical Trial Information Link (20 participants | 1 year | Ongoing)
Clinical Trial Information Link (US National Library of Medicine) (20 participants | 1 years | Ongoing)
Parkinson’s Disease
- Improvements in Clinical Signs of Parkinson’s Disease Using Photobiomodulation: A Prospective Proof-of-Concept Study
Institutes – University of Sydney, University of New South Wales, Griffith University
Published Study Link | Blog Summary | Device: Vielight Neuro Gamma - Pilot Intervention With Near Infrared Stimulation
Institutes – University of Florida
Clinical Trial Information Link (135 participants | 4 years | Ongoing) - Evaluation of Dose of Photobiomodulation (Light) Therapy and Physiotherapy for Improving Quality of Life Outcomes and Mobility in Parkinson’s Disease (Pilot)
Institutes – University of Sydney, University of Brisbane
Clinical Trial Information Link (Australian New Zealand Clinical Trial Registry) (16 participants | 1.5 years | Ongoing)
PTSD and Gulf War Illness
- Improvements in Gulf War Illness Symptoms After Near-Infrared Transcranial and Intranasal Photobiomodulation: Two Case Reports
Institutes – University of California San Francisco & Veterans Affairs USA
Published Study Link | Device: Vielight Neuro Alpha
Autism Spectrum Disorder (ASD)
- Transcranial Photobiomodulation for the Treatment of Children with Autism Spectrum Disorder (ASD): A Retrospective Study
Institutes – Istituto di Neuroscienze, Italy & Albert Einstein College of Medicine, NY
Published Study Link | Blog Summary | Device: Vielight Neuro Duo
Covid-19
- Vielight RX Plus for the Treatment of COVID-19 Respiratory Symptoms
Various clinical sites
Pre-print of Results (295 participants | 1 year | Pre-publication)
Medical Device License - Post-Covid – A Pilot Study Evaluating the Efficacy of the Vielight Neuro RX Gamma in the Treatment of Post COVID-19 Cognitive Impairment
Various clinical sites
Clinical Trial Information (36 participants | 1 year | Recruitment)
Brain EEG Modulation and Cellular Effects
- Enhancement of Divergent Creative Thinking After Transcranial Near-Infrared Photobiomodulation Over the Default Mode Network
Institutes – University of Deusto, University of Montpellier, Pennsylvania State University
Published Study Link | Device: Vielight Neuro Gamma - Near-Infrared Photobiomodulation of Living Cells, Tubulin, and Microtubules In Vitro
Institutes – University of Alberta, Princeton University, Politecnico di Torino, Vielight Inc
Published Study Link (Frontiers in Medical Technology, 2022) - Pulsed Near Infrared Transcranial and Intranasal Photobiomodulation Significantly Modulates Neural Oscillations: A Pilot Exploratory Study
Institutes – Centre for Therapeutic Brain Intervention, CAMH, Toronto
Published Study Link (Scientific Reports, 2019) | NCBI Link - Modulation of Cortical Oscillations Using 10Hz Near-Infrared Transcranial and Intranasal Photobiomodulation: A Randomized Sham-Controlled Crossover Study
Abstract Link (Brain Stimulation Journal, 2021)
Systemic, Anti-Viral and Anti-Microbial
- Vielight RX Plus for the Treatment of COVID-19 Respiratory Symptoms
Various clinical sites
Pre-print of Results (295 participants | 1 year | Pre-publication)
Medical Device License - Antimicrobial Photodynamic Inactivation Mediated by Tetracyclines in Vitro and in Vivo: Photochemical Mechanisms and Potentiation by Potassium Iodide
Institutes – Harvard Medical School
Published Study Link (2018) - Antimicrobial Blue Light Therapy for Infectious Keratitis: Ex Vivo and In Vivo Studies
Institutes – Harvard Medical School
Published Study Link (2017) - Antimicrobial Blue Light Inactivation of Candida albicans: In Vitro and In Vivo Studies
Institutes – Harvard Medical School
Published Study Link (2015)
If you are a researcher and interested in photobiomodulation research, we welcome you to reach out to us.
Our 7+ PhDs have collaborated with multiple organizations, including Harvard Medical School and the US VA for a decade.
The Vielight Neuro offers industry-leading energy density and an adjustable design to target various brain networks.
Please write to: [email protected]
What is near-infrared (NIR) light?
Near-infrared (NIR) light is a form of electromagnetic radiation with wavelengths between 700–1400 nanometers. It is mostly invisible to the naked eye, even at high power densities. NIR is widely studied for its ability to penetrate biological tissues and influence cellular processes — the foundational principle of photobiomodulation (PBM).
Do the devices come with instructions?
Yes. Every Vielight device includes a printed user manual in the packaging.
If you’ve misplaced it, digital copies are available here. (Insert link)
What conditions do the intranasal devices help with?
Vielight devices are currently classified as experimental and are not intended to diagnose, treat, or cure any condition.
However, our pilot study—conducted in collaboration with researchers from Harvard Medical School and Boston University School of Medicine—yielded statistically significant results.
To further validate our technology, we’re conducting clinical trials in partnership with respected institutions including:
- Baycrest Hospital (Canada)
- St. Michael’s Hospital (Canada)
- Veterans Affairs (USA)
How soon can I expect results?
Results vary significantly between individuals.
Some users report noticeable effects within a week, while others may take longer to perceive improvements in overall wellness and cognitive function.
Can I cover the intranasal applicator with plastic or other materials if I share it?
No. Covering the applicator (e.g., with plastic caps or wraps) will interfere with the coherence and effectiveness of the light output.
If sharing, we recommend sanitizing the applicator between uses or purchasing additional applicators.
Will the intranasal devices interfere with my pacemaker?
There are no known reports of Vielight devices interfering with pacemakers.
However, if you have concerns, consult your healthcare provider before use.
Can I bring and use Vielight devices on a plane?
Yes. Vielight devices are portable and safe for travel. They can be carried and used during flights.
Where are Vielight devices made?
Vielight devices are assembled in the USA and Canada, using components sourced globally.
Why 810nm vs 1064nm / 1070nm?
Vielight utilizes the 810nm (near-infrared) wavelength because it is scientifically recognized as the “sweet spot” for brain photobiomodulation. While competitors often market 1064nm and 1070nm as “superior” due to its longer wavelength, the peer-reviewed evidence favors 810nm for three critical reasons:
1. Superior Brain Penetration (The “Water Trap”)
Although longer wavelengths typically scatter less, wavelengths in the 1064nm and 1070nm range are more heavily absorbed by water. Since the brain is roughly 70–80% water, much of 1064nm and 1070nm photonic energy is absorbed by water molecules before it can reach deep brain tissue.
- The 810nm Advantage: 810nm sits in the “Optical Window” where absorption by water and blood is at its lowest, allowing it to penetrate the skull and reach the cortical tissue more efficiently.
2. Direct Mitochondrial Activation
The primary goal of brain PBM is to stimulate Cytochrome c Oxidase (CcO)—the “engine” of the mitochondria—to increase cellular energy (ATP).
- 810nm: Absorption by CcO peaks around 810nm. It directly triggers mitochondrial signaling.
- 1064nm / 1070nm: CcO absorption drops off significantly after 1000nm. Research suggests 1070nm works primarily through photothermal coupling (micro-heating water to open ion channels), which is a less direct and less studied path for cognitive enhancement.
3. The Evidence Leader
The 810nm wavelength is the most researched in the history of brain photobiomodulation.
- Published Data: Almost all major human clinical trials for Alzheimer’s, TBI, and PTSD use 810nm.
- Vielight’s Track Record: Vielight has over 25+ published studies and 15+ ongoing trials. In contrast, 1070nm has very few peer-reviewed human trials, meaning its long-term clinical effects are less understood.
4. High Irradiance Without Heat
To get light through the skull, you need high irradiance (power density). Because 810nm isn’t absorbed by water as much as 1070nm, Vielight devices can deliver powerful doses to the brain without causing uncomfortable or potentially harmful thermal stress (heat) on the scalp.
Summary: We use 810nm because it reaches the brain deeper, activates the mitochondria more effectively, and is supported by a mountain of clinical evidence that “borrowed” wavelengths simply do not have. Read more here: https://www.vielight.com/blog/810nm-vs-1070nm-wavelength-brain-photobiomodulation/
What if I don’t see any results?
If you’re unsatisfied with your results, Vielight offers a 6-month satisfaction guarantee with an 80% refund of the purchase price.
To initiate a return, please contact the original point of purchase.
⚠️ Important Note:
As of March 1, 2017, our customer support, satisfaction guarantee, and product warranty do not apply to devices purchased through unauthorized sellers, including Amazon and eBay.
What’s the difference between the Vielight 810 and the Neuro Alpha/Gamma?
- The Vielight 810 is a compact, intranasal-only device that uses near-infrared (NIR) light and is designed for everyday wellness and cognitive support.
- The Neuro Alpha and Neuro Gamma offer more powerful and comprehensive stimulation, combining a transcranial infrared headset with an intranasal applicator. These models deliver a significantly higher power density than the 810 and are designed for deeper brain photobiomodulation.
Recommended Usage:- Healthy cognitive function: Once every other day
- Noticeable cognitive decline: Once daily
📚 A full list of over 20 Vielight research and publications can be found here: vielight.com/research
What’s the difference between the Neuro Alpha and Neuro Gamma?
- Neuro Alpha:
Stimulates alpha brain wave activity, associated with mental calm, relaxation, and stress reduction.
→ View published abstract on Alpha wave research. - Neuro Gamma:
Stimulates gamma brain wave activity, correlated with focus, problem-solving, and cognitive activation.
→ Read peer-reviewed study on Gamma stimulation.
🔬 Please note: Vielight technologies are still under clinical investigation and are not approved by any national regulatory authority for treating specific medical conditions except Health Canada to “accelerate the recovery of upper respiratory symptoms in viral infections.“
What’s the difference between the Vielight 655 and 633?
- The Vielight 655 uses a low-level laser diode.
- The Vielight 633 uses a light-emitting diode (LED).
While lasers have been historically more studied in PBM, LEDs are gaining traction due to their safety profile, cost-effectiveness, and growing body of evidence.
I have the Vielight 810 but want to upgrade to the Neuro. What can I do?
If it has been less than 6 months since you purchased the Vielight 810, you’re eligible for an 80% refund under our satisfaction guarantee. This can be applied toward the purchase of a Neuro Alpha or Neuro Gamma.
Contact us to start the process.
Which intranasal unit is the best?
There’s no single “best” unit — the ideal device depends on your goals:
- Vielight 633 / 655:
Target systemic wellness, immune function, and cardiovascular health - Vielight 810 / Neuro Alpha / Neuro Gamma:
Focus on brain health, mental performance, and neurocognitive support
If you’re unsure which device fits your needs, reach out — we’re happy to help.
I don’t see any light in the 810 nm nose applicator. Is this normal?
Yes, this is completely normal. The 810 nm wavelength is in the near-infrared (NIR) range and is nearly invisible to the naked eye.
Some users mistakenly assume the device is faulty because the light appears dim or disappears when viewed at certain angles.
In reality, the power density is the strongest in the field of brain photobiomodulation, even though the light is barely visible. This is a key characteristic of therapeutic NIR light.
My device seems faulty. Where do I send it for repair or replacement?
If you suspect a fault, please contact us first so we can troubleshoot with you:
- 📞 North America: 1-877-355-8012
- 📧 Email: [email protected]
If the issue cannot be resolved and the device is still under warranty:
- Direct purchases: Return the unit to Vielight
- Authorized reseller purchases: Contact your reseller for return and replacement instructions
There’s no light from the intranasal applicator anymore. What should I do?
If your intranasal applicator isn’t lighting up, there may be two possible causes:
- The device was turned on before the applicator was connected
→ Turn the power off, attach the applicator securely, and then turn the device on again. - There’s a green light on the control unit labeled “APP Fault”
→ This indicates a faulty applicator. If your product is still under warranty, your supplier will replace it.
→ Out-of-warranty applicators can be purchased directly from vielight.com.
How long do the diodes last?
Vielight intranasal diodes are constructed using transparent high-impact polycarbonate and are engineered for long-term durability.
They are covered under a one-year manufacturer’s warranty.
What kind of batteries do Vielight devices use?
- Vielight MIP: Use 1.5V AA batteries
Do Vielight devices have FDA approval?
Vielight devices do not have FDA approval. Vielight technology is FDA registered under the Medical Specialty of Physical Medicine and is supported by our audited FDA QS820 compliant quality system. The FDA have also categorized our devices as low-risk general wellness devices in accordance to the FDA’s draft on “General Wellness: Policy on Low Risk Devices” – dated January 20, 2015. All Vielight devices have been independently TÜV certified as safe for consumer use.
Do Vielight devices have Health Canada approval?
The RX-Plus was used in a successful n=294 clinical trial for Covid-19 recovery.
We received a Health Canada Medical Device license that covers all upper respiratory symptoms from viral infections as follows, “The Vielight RX Plus is intended to accelerate the recovery of upper respiratory symptoms in viral infections, such as COVID-19”
- Clinical trial results can be found here: https://medrxiv.org/cgi/content/short/2022.06.16.22276503v1
- Information on the Health Canada license can be found here: https://health-products.canada.ca/mdall-limh/information?companyId=136815&lang=eng
Are Vielight devices covered by medical insurance?
Generally, no. Vielight devices are considered wellness and experimental technologies and are not covered by most insurance plans.
However, some private or alternative health insurance policies may cover wellness-related devices. We recommend checking with your provider.
However, some private or alternative insurance providers may offer reimbursement for wellness-related equipment. We recommend checking directly with your provider.
For U.S. Veterans:
Vielight has partnered with the Lovell Government Services to support Veterans’ access to wellness technologies. If you are a U.S. Veteran receiving care through VHA, you may be eligible for a Vielight device.
Learn more and apply here: vielight.com/veterans
What payment methods do you accept?
We currently accept the following payment methods:
- Visa
- MasterCard
- PayPal
What are your international shipping fees?
Our international shipping rates are standardized for most countries.
If your country does not appear during checkout, please contact us at [email protected] and we’ll provide specific shipping options and rates.
Will I need to pay additional customs duties or taxes?
Customs duties and import taxes depend on your country’s regulations.
- If you are located outside the USA, Canada, or Australia, you may be subject to additional charges upon delivery.
- We do take steps to mitigate customs-related costs where possible, but the final responsibility lies with the customer.
Do you offer a partnership program?
Yes — Vielight offers two active, non-exclusive partnership programs:
- Reseller Program: For businesses interested in selling Vielight devices.
- Affiliates Program: For individuals or organizations who wish to refer others and earn commissions.
These programs are ideal for health professionals, wellness advocates, and anyone passionate about photobiomodulation.
🔗 Learn more here: vielight.com/business-partners
🩺 For Practitioners:
We also offer a dedicated program for clinicians and healthcare professionals.
Explore the benefits: vielight.com/practitioners-program
How do you ship?
We ship globally to most countries. It takes 5 to 7 business days for your purchase to reach its destination, once your order has been processed.
We ship via local postal services within Canada, Australia, and the United States, and internationally using private courier. Vielight does not charge taxes, but your local country’s customs agencies will reach out for any import taxes that they may be required to pay, and we are unable to waive any of these fees. The average shipping time for international orders may be longer depending on inspections by your local customs agency.
What are the shipping requirements for each country?
USA: Deliveries of all Neuro, XPlus, Vagus, and other products with a power bank require an Adult Restricted Delivery Signature, where only the receiver can sign for delivery and proof of ID is required.
Canada: Deliveries to all Canadian addresses require presentation of an ID card at pick-up.
Australia: Deliveries to all Australian addresses require a delivery signature.
Outside of Australia, Canada, and the USA: Delivery Signature and payment of customs duty are required.
What is your return and refund policy?
We offer an 80% refund within 6 months of purchase if you’re not satisfied with your Vielight device.
This applies only to:
- Direct purchases from Vielight, or
- Purchases from authorized global resellers
To request a refund, please contact the point of purchase directly.
🔒 Note: Intranasal applicators purchased separately are non-refundable.
Is there a product warranty?
Yes. All Vielight devices are backed by a 2-year warranty against defects in materials and workmanship.
If your device is found to be defective within this period, we will repair or replace it at no charge.
Important Notice on Unauthorized Sellers
We do not support or honor warranty/refund claims for devices purchased through unauthorized third-party sellers, including:
- Amazon
- eBay
- Other online marketplaces
These products may be counterfeit, stolen, or tampered with.
⚠️ Genuine Vielight devices bear the official “Vielight” logo and are only available through Vielight.com or our authorized resellers.
If you’re unsure whether your device is authentic, please contact us for verification.
Do you charge any hidden or additional fees?
No. Neither Vielight nor our payment processor charges any fees beyond the total purchase amount shown at checkout.
However, some credit card issuers may add foreign exchange or international transaction fees. These charges are imposed by your bank and are not controlled by Vielight.