Vielight Improves Brain Resilience: Published Neuroprotection Research | Football
Note on Research: The study discussed in this article was a peer-reviewed, double-blind, sham-controlled exploratory investigation published in the Journal of Neurotrauma. While the findings are statistically significant and promising, the authors note that the results should be considered preliminary and hypothesis-generating. Further research, including larger multi-site trials, is warranted to fully establish clinical efficacy for this specific application.
Read the full published study in the Journal of Neurotrauma here: https://journals.sagepub.com/doi/epub/10.1177/08977151251403554
Read the University of Utah’s press release here: https://healthcare.utah.edu/newsroom/news/2026/01/medical-grade-near-infrared-light-therapy-specific-frequencies-shows-promise
For decades, the conversation around American football and brain health has been dominated by a sense of inevitability. We know about the risks of Chronic Traumatic Encephalopathy (CTE). We know that it’s not just the “big hits” (concussions) that matter, but the thousands of “micro-hits” known as Repetitive Head Acceleration Events (RHAE) that accumulate over a season.
Until now, our best defense has been reactive. But what if we could make the brain itself more resilient to the hit before it even happens?
A groundbreaking new study with the Vielight Neuro by the University of Utah provides the first evidence that we might be able to. Published this month in the Journal of Neurotrauma—a top-tier, Q1 journal and the official publication of the National and International Neurotrauma Societies—the study offers a rigorous validation of a new approach.
The secret weapon? A specific, medical-grade application of light therapy known as itPBM (intranasal-transcranial photobiomodulation).
The Study by the Numbers
Lead author Dr. Hannah Lindsey and senior author Dr. Elisabeth Wilde recruited 26 active NCAA Division I collegiate football players for this randomized, double-blind, sham-controlled trial.
The players were split into two groups and monitored over a full 16-week season (preseason through regular season).
-
Active PBM Group (n=13): Self-administered the therapy 3 days a week for 20 minutes per session.
-
Sham PBM Group (n=13): Wore an identical device that delivered no therapeutic light.

The Vielight Neuro 3, superseded by the Vielight Neuro 4
The Technology: 40 Hz Gamma and the "Nasal Key"
This study utilized the Vielight Neuro Gamma 3 (superseded by the Neuro Gamma 4), a specialized device designed to deliver itPBM—a combination of intranasal and transcranial photobiomodulation.
The specific technical parameters were crucial to the study’s design:
-
Wavelength: 810 nm (Near-Infrared), chosen for its ability to penetrate deeper into tissue.
-
Pulse Rate: 40 Hz, a frequency associated with gamma brain oscillations.
-
Power Density: 100 mW/cm² for the transcranial LEDs and 25 mW/cm² for the intranasal applicator.
-
Total Dose: A substantial energy delivery of 240 J/cm² per session.
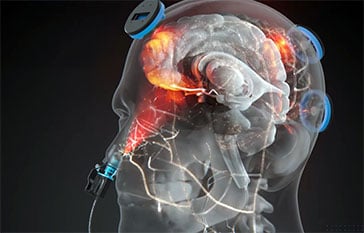
Why the Nose Matters: The device includes an intranasal LED positioned to illuminate the olfactory bulbs and orbitofrontal cortex. This “pincer maneuver”, combining transcranial LEDs on the skull with an intranasal applicato, allows light to reach ventral (underside) brain structures often missed by standard headsets.
To measure the effects, the researchers used advanced diffusion MRI to track two specific biomarkers:
-
Restricted Diffusion Imaging (RDI): A highly sensitive marker for neuroinflammation (immune cell infiltration).
-
Quantitative Anisotropy (QA): A marker for axonal remodeling and structural repair.
The statistical divergence between the two groups was massive.

The sham PBM group showed increases in diffusion imaging (RDI) across white-matter tracts from preseason to postseason. The active PBM group displayed decreases in selected tracts.
The Findings: "Fire vs. Ice" in the Data
1. The Sham Group (No Light): A Brain on Fire
Without protection, the impacts of the season took a heavy toll.
-
RDI (Inflammation): Showed a statistically significant increase (T=3.15, FDR=0.000) with a very large effect size (R²=0.50). This indicates that 50% of the variance in inflammation was driven by the cumulative head impacts over time.
-
QA (Axonal Stress): Similarly increased (T=2.70, FDR=0.000, R²=0.42), suggesting the brain was actively trying to remodel damaged axons.
2. The Active itPBM Group: Neuroprotective Stability
Despite taking the same hits and playing the same game, the treated athletes showed a completely different brain profile.
-
Relative Stability: The Active group showed no significant increases in inflammation or axonal remodeling markers.
-
Signs of Recovery: In some regions, the Active group actually showed statistically significant decreases in RDI (T=0.77, FDR=0.015), suggesting the therapy may have actively facilitated recovery processes during the season.

Protecting the "Cone of Vulnerability"
One of the most scientifically robust findings was the location of the protection. The study identified that the Sham group’s injury was concentrated in the “Cone of Vulnerability”—deep brain regions most susceptible to the rotational forces of a tackle.
Specifically, the Sham group showed significant damage in:
-
The Midbrain and Brainstem.
-
The Corpus Callosum (Forceps Major and Minor).
-
The Thalamus and Basal Ganglia.
In the Active itPBM group, these exact deep-structure regions were spared. This suggests that the intranasal component of the therapy successfully delivered therapeutic light to the brain’s deepest, most vulnerable survival centers.

An Era of Brain Resilience
The implications of this research are massive. We are moving from a paradigm of “investigating concussions” to “prophylactic neuroprotection”.
As the authors noted, current strategies like helmets reduce force but do not treat the cellular cascade of injury. This study provides the first evidence that itPBM can intervene at the cellular level, mitigating the neuroinflammatory response (RDI) before it spirals into long-term damage.
For athletes, this data suggests that medical-grade itPBM could become as essential to their routine as weight training—a non-invasive way to “armor” the brain from the inside out.
What is itPBM—and how is it different from transcranial-only PBM (tPBM)?
Intranasal-transcranial photobiomodulation (itPBM) combines transcranial photobiomodulation with intranasal brain photobiomodulation. The intranasal channel is a direct channel to the brain’s deepest regions, located on the underside of the brain, such as the hypothalamus and ventromedial prefrontal cortex.
While our industry leading Vie-LED transcranial LEDs cover the top of the brain, our patented intranasal module completes the circuit from the underside, covering both the top and bottom sides of the brain – creating patented, comprehensive coverage of the brain that other tPBM-only devices cannot match.
Vielight intranasal technology has been clinically proven to be 20x more effective than transcranial-only helmets in an MRI-based n=45 clinical study by Baycrest Hospital.
What did the MRI actually measure (RDI and QA)?
The researchers used advanced diffusion MRI metrics to track changes in white-matter microstructure across the season. They discuss RDI as a marker that can align with neuroinflammatory cellular processes in this context, and QA as a metric related to tract integrity/remodeling.
Does this prove PBM prevents concussion, CTE, or long-term brain disease?
No. This study suggests a promising signal worth validating in larger trials, but it does not prove prevention of disease.
Researchers at the University of Utah and NYU have received a landmark $4.6 million grant from the U.S. Department of Defense to investigate Vielight’s photobiomodulation (PBM) technology as a treatment for traumatic brain injury (TBI).
This large-scale, 300-participant clinical trial aims to establish a drug-free intervention for service members and first responders suffering from chronic mTBI symptoms. Led by Dr. Elisabeth Wilde and Dr. Carrie Esopenko, the study will examine the efficacy of Vielight technology in improving cognitive function, mood regulation, and overall brain health.
This is based on several successful medium sized clinical trials (40+ participants) with Vielight’s patented intranasal-transcranial technology.
Why 810 nm and 40 Hz?
The protocol used 810 nm near-infrared light because it’s a well-studied PBM wavelength with favorable tissue penetration and a long track record in neurological research applications. The light was pulsed at 40 Hz (gamma range) rather than delivered continuously, since pulsing can change the biological “dose profile” and may influence network-level responses differently from steady illumination. Importantly, this paper reports results for this exact parameter set—it doesn’t prove that other wavelengths or pulse rates would produce the same outcomes.
Sources:
-
Lindsey, H. M., et al. (2026). Transcranial Photobiomodulation Promotes Neurological Resilience in Current Collegiate American Football Players Exposed to Repetitive Head Acceleration Events. Journal of Neurotrauma.