Updates
Vielight Enters New Research Frontier in Japan: Schizophrenia Research
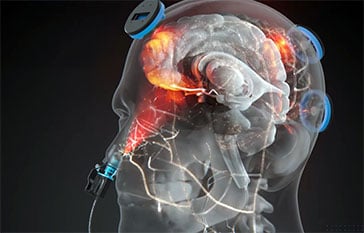
Clinical Protocol Expansion The 12-week open-label trial (jRCT032240756) is designed to evaluate whether itPBM can safely mitigate Cognitive Impairment Associated with Schizophrenia (CIS). Participants undergo 20-minute sessions three times per week using a multi-site system that combines an intranasal applicator with five transcranial scalp sensors – targeting the frontal, vertex, temporal, and occipitoparietal regions. This […]