Photobiomodulation
Learn about the science behind Vielight devices, why irradiance and wavelengths matter, and what makes photobiomodulation therapy so effective for your brain.
How photobiomodulation therapy boosts your brain's potential

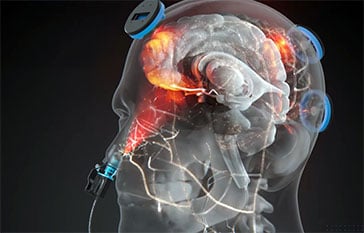
Near-infrared (NIR) energy penetrates the skull

The NIR energy reaches brain cells

Mitochondria absorb NIR energy, boosting cellular energy and brain function
Photobiomodulation benefits
Increases the production of Adenosine Triphosphate (ATP)
A molecule that fuels nearly all biological activity, including cellular growth and repair.
Promotes the release of
Nitric Oxide (NO)
A molecule that enhances cellular signaling and improves blood circulation.
Generates low levels of Reactive Oxygen Species (ROS)
Mild oxidants that trigger gene transcription for cellular repair and healing.
Wavelength x Irradiance

What is wavelength and irradiance?
In photobiomodulation, the concentration of light energy (irradiance) is vital besides wavelength. A systematic review of over 2000+ PBM studies found the average parameters for effective therapy were 810nm and 250 mW/cm².
Vielight’s Vie-LED technology delivers high-intensity, laser-like energy, surpassing the weaker irradiance of helmet-style devices.
Even with hundreds of LEDs, helmets dilute power by sharing electrical current across many diodes. This weak irradiance prevents deep penetration, reducing effectiveness.


Energy Footprint Comparison on
a real human skull


The Science Behind Vielight


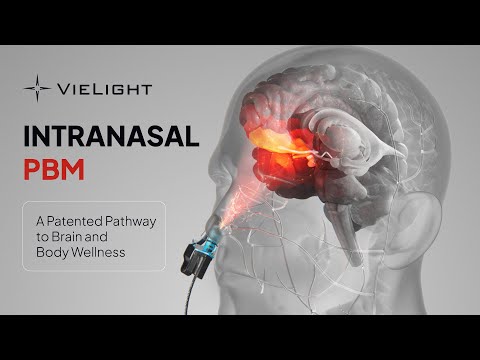
What is brain photobiomodulation?
The brain, our most complex organ, relies on mitochondria in each cell as energy-producing “batteries.”
Photobiomodulation improves mitochondrial performance. This boost in cellular energy helps heal brain cells, repair neuron connectivity, and improve brain efficiency, ultimately enhancing overall brain function.
Near-infrared light, especially at 810 nm, penetrates deepest into brain tissue. To fine-tune photobiomodulation parameters for optimal results, research that uses Vielight technology employs brain imaging and brain signaling techniques.
What is the Vielight Neuro 4?

Published research with Vielight technology
Explore peer-reviewed studies featuring Vielight technology in collaboration with world-class institutions.