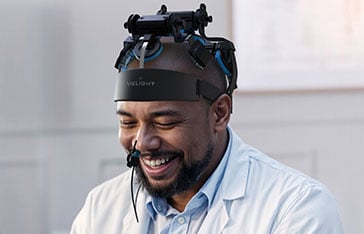
Vielight Enters New Research Frontier in Japan: Schizophrenia Research
TOKYO, JAPAN – Researchers at the National Center of Neurology and Psychiatry have announced a new clinical trial protocol exploring the use of Vielight intranasal-transcranial photobiomodulation (itPBM) for patients with schizophrenia.
Published in the peer-reviewed journal Neuropsychiatric Disease and Treatment, the study protocol marks a pivotal shift in how “General Wellness” light therapy devices are being evaluated for complex psychiatric conditions.
Key Highlights of the Study:
Focus on Cognition: The trial specifically targets Cognitive Impairment Associated with Schizophrenia (CIS)
Dual-Delivery Method: Vielight Neuro intranasal-transcranial PBM
Clinical Rigor: Improvements in social functioning, activities of daily living, and cerebral blood flow.
Clinical Protocol Expansion
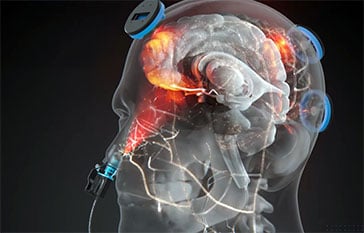
The 12-week open-label trial (jRCT032240756) is designed to evaluate whether itPBM can safely mitigate Cognitive Impairment Associated with Schizophrenia (CIS). Participants undergo 20-minute sessions three times per week using a multi-site system that combines an intranasal applicator with five transcranial scalp sensors – targeting the frontal, vertex, temporal, and occipitoparietal regions. This specific configuration is intended to synchronize neural oscillations and enhance cerebral blood flow, addressing the “misfiring” or diminished gamma activity often observed in patients with schizophrenia.
To measure success, the researchers are employing a rigorous battery of primary and secondary endpoints. Beyond basic safety and tolerability monitoring over a six-month period, the study tracks improvements in neurocognitive function through the WAIS Digit Symbol Coding and Rey Auditory Verbal Learning tests. Furthermore, it explores the impact on daily life using the Specific Levels of Functioning (SLOF) scale and monitors physiological changes in the brain via near-infrared spectroscopy (NIRS) and event-related potentials (ERPs).
Participating Institutions
The research is being spearheaded by the National Center of Neurology and Psychiatry (NCNP) in Tokyo, Japan. Established in 1986 as one of Japan’s six national centers, the NCNP is a globally recognized hub for integrated brain research. It uniquely bridges the gap between laboratory science and patient care by housing the National Institute of Mental Health and the National Center Hospital on a single campus. This close-knit structure allows the Department of Preventive Intervention for Psychiatric Disorders to translate cutting-edge technology, like photobiomodulation, directly into clinical protocols for complex conditions.
Collaborating on this initiative is the Japan Health Research Promotion Bureau, an organization dedicated to streamlining clinical innovation across the country. The study is supported by a Grant-in-Aid for Scientific Research from the Japanese government, underscoring the high priority placed on finding non-pharmacological solutions for psychiatric rehabilitation. By involving the NCNP’s Department of Clinical Data Science, the trial ensures that the preliminary safety data and biomarker results are analyzed with the highest level of academic and statistical rigor.