Utah Phase 2 TBI Study | Cognitive Gains Reported with Vielight Neuro Gamma
Educational summary of published clinical research on intranasal-transcranial photobiomodulation (itPBM) using Vielight technology in individuals with a history of repetitive head acceleration events (RHAEs) and related cognitive concerns.
The Vielight Neuro is categorized as a general wellness device by the FDA. It is not cleared or approved by the U.S. FDA to diagnose, treat, cure, mitigate, prevent, or manage traumatic brain injury (TBI).
Investigating Photobiomodulation as a Cognitive Intervention for Repetitive Head Acceleration Events
This TBI clinical study conducted by researchers from the University of Utah, Brigham Young University, and affiliated institutions explored the potential of intranasal-transcranial photobiomodulation (itPBM) with the Vielight Neuro Gamma to improve cognitive function in individuals with a history of repetitive head acceleration events (RHAEs).
This U of Utah and Vielight TBI study (n=44) is the largest published human study to report statistically significant improvements on objective cognitive tests after a multi-week PBM intervention in a TBI-related/RHAE population. Larger trials exist, but they did not demonstrate significant clinical (symptom/cognition) gains.
This builds on the previous n=43 published TBI clinical study by the University of Utah.
These events, often experienced in contact sports and military contexts, may not cause immediate concussions but are known to contribute cumulatively to cognitive decline and increased risk of neurodegeneration over time.
READ THE FULL PUBLISHED STUDY HERE | Published Study Link
“Football almost killed me… But Vielight saved my life.” — Dr. Larry Carr.
Study Objective
The goal was to assess whether near-infrared PBM therapy using an 810 nm LED device could produce measurable improvements in cognitive performance. This approach was motivated by the need for effective, non-invasive treatments targeting the underlying neural mechanisms affected by RHAEs.
Participant Profile
-
N = 44 (90% male; mean age = 46)
-
History of RHAEs averaging 12.4 years
-
Participants were excluded if they had known neurological diseases or major psychiatric disorders
Intervention Protocol
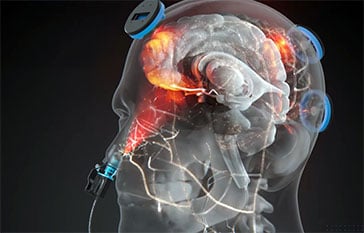
Each participant received a Vielight Neuro Gamma (v3) device, which delivers near-infrared light to cortical and subcortical brain regions through both transcranial and intranasal routes:
-
LEDs targeted the dorsomedial prefrontal cortex, lateral parietal lobes, and midline precuneus
-
An intranasal LED directed light toward orbitofrontal and olfactory-associated brain structures
-
Sessions lasted 20 minutes and were conducted every other day over 8–10 weeks (mean = 29 sessions)

Cognitive Assessments
A comprehensive battery of validated neuropsychological tests was administered before and after the intervention, measuring domains including:
-
Verbal learning and memory (CVLT-3)
-
Executive function (D-KEFS)
-
Sustained attention (CPT-3)
-
Global cognition (NIH Toolbox Cognition Battery)

Key Findings
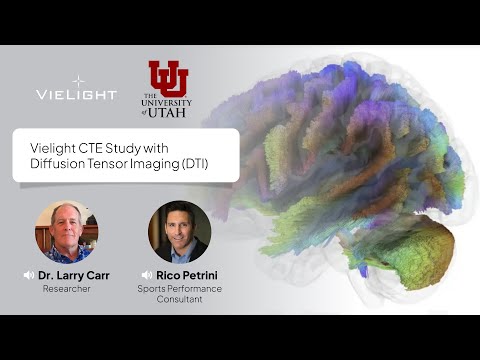
University of Utah diffusion-MRI (DTI) tractography maps show decreased inflammation-related diffusion markers with Vielight Neuro versus placebo
Group-level improvements were statistically significant across several domains:
-
Verbal memory: Learning and delayed recall improved (Cohen’s d = 0.49–0.62)
-
Executive function: Inhibition and cognitive switching improved (d = 0.54–0.67)
-
Attention: Enhanced sustained attention and fewer omission/commission errors (d = -0.34 to -0.67)
-
Fluid cognition and processing speed: Moderate-to-large effect sizes observed (d = 0.45–0.94)
Crystallized abilities such as vocabulary and reading remained stable, supporting the specificity of PBM’s effect on vulnerable cognitive functions.
Individual-Level Analysis
Reliable Change Index (RCI) calculations revealed:
-
14–36% of participants showed reliable improvement in select domains
-
Improvements were most common in attention, memory, and processing speed
-
Very few participants demonstrated any reliable cognitive decline
Proposed Mechanism
PBM is believed to act via mitochondrial cytochrome c oxidase, leading to increased ATP production, nitric oxide release, anti-inflammatory effects, and neuroplastic adaptations. These mechanisms may underlie the observed cognitive benefits.
Conclusion
This study provides preliminary evidence that PBM may offer cognitive benefits for individuals exposed to repetitive neurotrauma. The observed improvements, particularly in fluid cognition and memory, suggest PBM could be a promising candidate for non-pharmacological neurorehabilitation. While further research is needed to validate these findings, this study marks an important step toward establishing PBM as a viable intervention for cognitive impairment following RHAEs.