Long COVID-Brain Fog Clinical Trial: Vielight Neuro (The Lancet)
Link to published study in The Lancet eClinical Medicine: https://www.thelancet.com/journals/lanepe/article/PIIS2589-5370(25)00665-0/fulltext
This article is for educational purposes only and summarizes findings from a published randomized, sham-controlled pilot trial in adults with post-COVID condition–related cognitive symptoms (“brain fog”). The Vielight Neuro is categorized as a general wellness device by the FDA. The Vielight Neuro is not cleared by the FDA to treat, cure or prevent Long COVID-related brain fog.
Toronto, Canada — January 13, 2026
A new randomized, double-blind, sham-controlled clinical trial with 43 participants reports that the Vielight Neuro produced measurable improvements in objective cognitive performance in adults under 45 years old with long COVID-related brain fog. This is often a disabling condition that continues to limit workforce participation, strain healthcare systems, and undermine mental well-being worldwide.
Cognitive dysfunction, often referred to as “brain fog,” is one of the most persistent and debilitating symptoms of post-COVID-19 condition (PCC). It interferes with attention, memory, and executive function, limiting the ability of affected individuals, many of them of working age, to return to work, education, and daily responsibilities. Beyond the personal toll, long COVID brain fog contributes to reduced workforce participation, increased healthcare utilization, and rising mental-health burden, creating ripple effects across the broader economy and society.

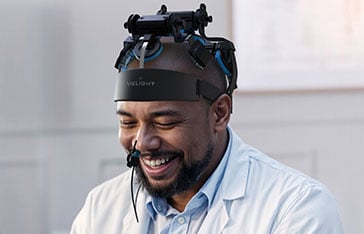
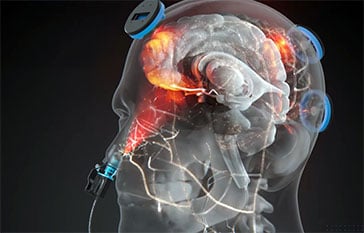
Figure. The intervention itPBM device, Vielight Neuro RX Gamma and targeted hubs of the default mode network (DMN) and cerebellum.
The study, published today in The Lancet’s eClinicalMedicine, evaluated intranasal and transcranial photobiomodulation (itPBM), a form of near-infrared light stimulation designed to support brain metabolism and network function. The intervention was delivered using the Vielight Neuro RX Gamma, a itPBM device configured for this clinical trial. The trial represents one of the most rigorously controlled investigations to date of a scalable, home-use intervention targeting cognitive dysfunction associated with long COVID.
Addressing a Major Unmet Need
Despite affecting millions globally, there are currently no established, evidence-based treatments specifically approved for PCC-related cognitive impairment. Many patients report being left without therapeutic options, relying instead on symptom management and coping strategies.
“This is a major unmet medical and societal need,” said Dr. Lew Lim, corresponding author of the study and founder of Vielight Inc. “Long COVID brain fog doesn’t just affect individuals. It impacts families, employers, healthcare systems, and mental well-being. Our objective was to rigorously evaluate whether a non-invasive, home-based intervention could produce objective signals of cognitive improvement, particularly in people still early in their working lives.”

Figure: Primary endpoint at Day 56 (Active–Sham), stratified by age. Least-squares mean difference favored the active group in participants <45 years (p=0.028)
Study Design and Key Findings
The U.S.-based pilot trial enrolled 43 adults aged 18-65 with persistent cognitive symptoms at least 12 weeks following COVID-19 infection. Participants were randomized to receive either active itPBM or a visually identical sham intervention for eight weeks, with participants, investigators, and outcome assessors blinded to treatment allocation.
Unlike many long COVID studies that rely primarily on self-reported symptoms, the primary endpoint was objective cognitive performance, assessed using a validated digital cognitive battery measuring attention, memory, and executive function.
Key findings included:
- Participants under 45 years of age receiving active itPBM showed statistically significant improvements in composite cognitive performance compared with sham.
- Tasks related to attention demonstrated consistent signals of improvement across multiple timepoints.
- The intervention was well tolerated, with no serious adverse events, and adherence to unsupervised home use was high.
- In the full study population, participants receiving active treatment showed greater improvement than sham at the primary endpoint, although the overall between-group difference did not reach conventional statistical significance.
- Secondary outcomes, including fatigue and quality-of-life measures, showed mixed results, reflecting the heterogeneity of long COVID.